 This group elements are also called as halogens. 
These elements have seven electrons in their valence shell. Their general electronic configuration is ns 2np 5. This group elements are also known as chalcogens.įluorine, Chlorine, Bromine, Iodine and Astatine are the elements of this group. 
These elements have six electrons in their valence shell. Their general electronic configuration is ns 2np 4. Oxygen, Sulphur, Selenium, Tellurium and Polonium are the elements of this group. These group elements are also called as pnictogens. These elements have five electrons in their valence shell. Their general electronic configuration is ns 2np 3. Nitrogen, Phosphorous, Arsenic, Antimony and Bismuth are the elements of this group. These elements have four electrons in their valence shell. 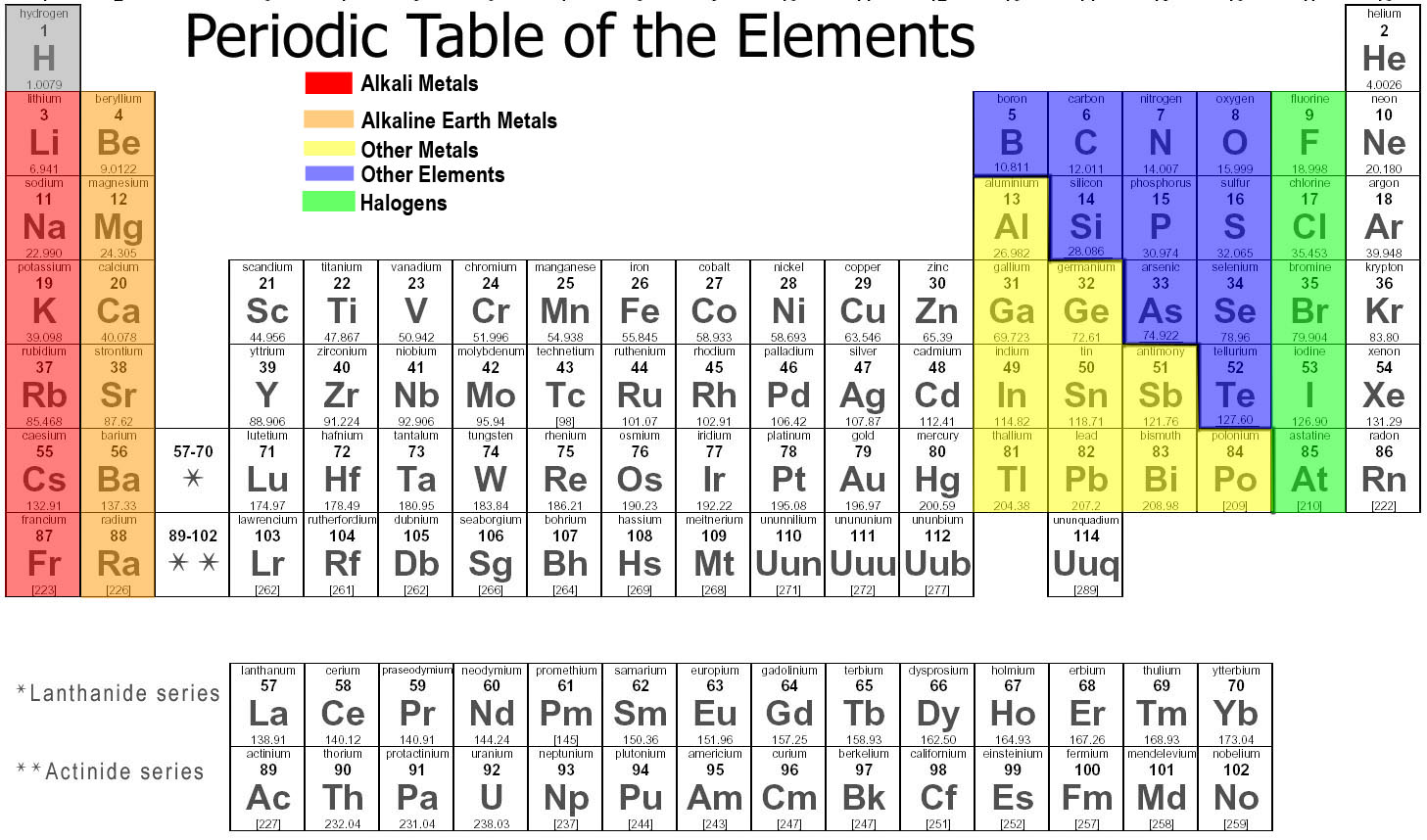
Their general electronic configuration is ns 2np 2. These group elements mostly form covalent compounds.Ĭarbon, Silicon, Germanium and Tin are the elements of this group. These elements have three electrons in their valence shell. Their general electronic configuration is ns 2np 1. These elements obtained from earth crust because of these reasons these elements are called as alkaline earth metals.īoron, Aluminium, Gallium, Indium and Thallium are the elements of this group. The hydroxides of these group elements are basic in nature and soluble in water. These elements have two electrons in their valence shell. Their general electronic configuration is ns 2. This group elements are also known as alkali metals as the hydroxides of these group elements are basic in nature and soluble in water.īeryllium, Magnesium, Calcium, Strontium, Barium and Radium are the elements of this group. These elements have one electron in their valence shell. Their general electronic configuration is ns 1. Lithium, Sodium, Potassium, Rubidium, Cesium and Francium are the elements of this group. The elements of group -1, group-2 and group-13 to group-17 are the representative elements. The elements in the modern periodic table were classified into four types.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
